NEET AIPMT Chemistry Chapter Wise Solutions – s-Block Elements
Contents
NEET AIPMT Chemistry Chapter Wise SolutionsPhysicsBiology
1. Ionic mobility of which of the following alkali metal ions is lowest when aqueous solution of their salts are put under an electric field? (NEET 2017)
(a) K
(b) Rb
(c) Li
(d) Na
2. The suspension of slaked lime in water is known as (NEET-11 2016)
(a) lime water
(b) quick lime
(c) milk of lime
(d) aqueous solution of slaked lime.
3. In context with beryllium, which one of the following statements is incorrect? (NEET-II2016)
(a) It is rendered passive by nitric acid.
(b) It forms Be2C.
(c) Its salts rarely hydrolyze.
(d) Its hydride is electron-deficient and polymeric.
4. Which of the following statements is false? (NEET-I 2016)
(a) Ca2+ ions are not important in maintaining the regular beating of the heart.
(b) Mg2+ ions are important in the green parts of the plants.
(c) Mg2+ ions form a complex with ATP.
(d) Ca2+ ions are important in blood clotting.
5. The product obtained as a result of a reaction of nitrogen with CaC2 is (NEET-I 2016)
(a) CaCN3
(b) Ca2CN
(c) Ca(CN)2
(d) CaCN
6. On heating which of the following releases C02 most easily? (2015)
(a) Na2CO3
(b) MgCO3
(c) CaC03
(d) K2CO3
7. 20.0 g of a magnesium carbonate sample decomposes on heating to give carbon dioxide and 8.0 g magnesium oxide. What will be the percentage purity of magnesium carbonate in the sample?
(At. wt. of Mg = 24) (2015)
(a) 96
(b) 60
(c) 84
(d) 75
8. The function of “Sodium pump” is a biological process operating in each and every cell of all animals. Which of the following biologically important ions is also a constituent of this pump? (2015, Cancelled)
(a) K+
(b) Fe2+
(c) Ca2+
(d) Mg2+
9. Solubility of the alkaline earth metal sulphates in water decreases in the sequence (2015, Cancelled)
(a) Sr>Ca>Mg>Ba
(b) Ba>Mg>Sr>Ca
(c) Mg>Ca>Sr>Ba
(d) Ca > Sr > Ba > Mg
10. In Castner- Kellner cell for production of sodium hydroxide (Karnataka NEET 2013)
(a) brine is electrolyzed using graphite electrodes
(b) molten sodium chloride is electrolysed
(c) sodium amalgam is formed at mercury cathode
(d) brine is electrolyzed with Pt electrodes
11. Which one of the alkali metals, forms only, the normal oxide, M2O on heating in air? (2012)
(a) Rb
(b) K
(c) Li
(d) Na
12. The ease of adsorption of the hydrated alkali metal ions on an ion-exchange resins follows the order: (2012)
(a) Li+< K+< Na+< Rb+
(b) Rb+ < K+ < Na+ < Li+
(c) K+< Na+ < Rb+ < Li+
(d) Na+ < Li+ < K+ < Rb+
13. Which of the following compounds has the lowest melting point? (2011)
(a) CaCl2
(b) CaBr2
(c) Cal2
(d) CaF2
14. Which one of the following is present as an active ingredient in bleaching powder for bleaching action? (2011)
(a) CaOCl2
(b) Ca(OCl)2
(c) CaO2Cl
(d) CaCl2
15. Match List-I with List-II for the compositions of substances and select the correct answer using the code given above. (Mains 2011)
(a) (A)-(iii), (B)-(iv), (C)-(i), (D)-(ii)
(b) (A)-(ii), (B)-(iii), (C)-(iv), (D)-(i)
(c) (A)-(i), (B)-(ii), (C)-(iii), (D)-(v)
(d) (A)-(iv), (B)-(iii), (C)-(ii), (D)-(i)
16. Which of the following alkaline earth metal sulphates has hydration enthalpy higher than the lattice enthalpy? (2010)
(a) CaS04
(b) BeS04
(c) BaS04
(d) SrS04
17. Property of the alkaline earth metals that increases with their atomic number (2010)
(a) solubility of their hydroxides in water
(b) solubility of their sulphates in water
(c) ionization energy
(d) electronegativity
18. Which one of the following compounds is a peroxide? (2010)
(a) K02
(b) BaO2
(c) Mn02
(d) N02
19. The compound A on heating gives a colourless gas and a residue that is dissolved in water to obtain B. Excess of C02 is bubbled through aqueous solution of B, C is formed which is recovered in the solid form. Solid C on gentle heating gives back A. The compound is (Mains 2010)
(a) CaC03
(b) Na2C03
(c) K2C03
(d) CaS04-2H20
20. In the case of alkali metals, the covalent character decreases in the order (2009)
(a) MF > MCI > MBr > MI
(b) MF > MCI > Ml > MBr
(c) MI > MBr > MCI > MF
(d) MCI > MI> MBr > MF
21. Which of the following oxides is not expected to react with sodium hydroxide? (2009)
(a) CaO
(b) Si02
(c) BeO
(d) B203
22. Equimolar solutions of the following were prepared in water separately. Which one of the solutions will record the highest pH? (2008)
(a) MgCl2
(b) CaCl2
(c) SrCl2
(d) BaCl2
23. The sequence of ionic mobility in aqueous solution is (2008)
(a) Rb+ > K+ > Cs+ > Na+
(b) Na+ > K+ > Rb+ > Cs+
(c) K+> Na+> Rb+ > Cs+
(d) Cs+> Rb+ > K+ > Na+
24. The alkali metals form salt-like hydrides by the direct synthesis at elevated temperature. The thermal stability of these hydrides decreases in which of the following orders? (2008)
(a) NaH > LiH > KH > RbH > CsH
(b) LiH > NaH > KH > RbH > CsH
(c) CsH > RbH > KH > NaH > LiH
(d) KH > NaH > LiH > CsH > RbH
25. The correct order of increasing thermal stability of K2C03, MgC03, CaC03 and BeC03is (2007)
(a) BeC03 < MgC03< CaC03 < K2C03
(b) MgC03 <BeC03 < CaC03 < K2C03
(c) K2C03 < MgC03< CaC03 < BeC03
(d)BeC03 < MgC03 <K2C03< CaC03
26. In which of the following the hydration energy is higher than the lattice energy? (2007)
(a) MgS04
(b) RaS04
(c) SrS04
(d) BaS04
27. The correct order of the mobility of the alkali metal ions in aqueous solution is (2006)
(a) Rb+> K+> Na+> Li+
(b) Li+> Na+ > K+> Rb+
(c) Na+ > K+ > Rb+ > Li+
(d) K+> Rb+ > Na+ > Li+
28. The correct sequence of increasing covalent character is represented by (2005)
(a) LiCl < NaCl < BeCL2
(b) BeCl2< LiCl < NaCl
(c) NaCl < LiCl < BeCl2
(d) BeCl2 < NaCl < LiCl
29. A solid compound X on heating gives C02 gas and a residue. The residue mixed with water forms Y. On passing an excess of C02 through Y in water, a clear solution Z is obtained. On boiling Z, compound X is reformed. The compound X is (2004)
(a) Ca(HC03)2
(b) CaC03
(c) Na2C03
(d) K2C03
30. In which of the following processes, fused sodium hydroxide is electrolysed at a 330°C temperature for extraction of sodium? (2000)
(a) Castner’s process
(b) Down’s process
(c) Cyanide process
(d) Both ‘b’ and ‘c
31. When a substance (A) reacts with water it produces a combustible gas (B) and a solution of substance (C) in water. When another substance (D) reacts with this solution of (C), it also produces the same gas (B) on warming but (D) can produce gas (B) on reaction with dilute sulphuric acid at room temperature. Substance (A) imparts a deep golden yellow colour to a smokeless flame of Bunsen burner. Then (A), (B), (C and (D) respectively are (1998)
(a) Ca, H2, Ca(OH)2, Sn
(b) K, H2, KOH, A1
(c) Na, H2, NaOH, Zn
(d) CaC2, C2H2, Ca(OH)2, Fe
32. Calcium is obtained by the (1997)
(a) electrolysis of solution of calcium chloride in water
(b) electrolysis of molten anhydrous calcium chloride
(c) roasting of limestone
(d) reduction of calcium chloride with carbon.
33. Sodium is made by the electrolysis of a molten mixture of about 40% NaCl and 60% CaCl2 because (1995)
(a) Ca++ can reduce NaCl to Na
(b) Ca++ can displace Na from NaCl
(c) CaCl2 helps in conduction of electricity
(d) this mixture has a lower melting point than NaCl.
34. The solubility in water of sulphate down the Be group is Be > Mg > Ca > Sr > Ba. This is due to (1995)
(a) decreasing lattice energy
(b) high heat of solvation for smaller ions like Be2+
(c) increase in melting points
(d) increasing molecular weight.
35. Identify the correct statement. (1995)
(a) Plaster of Paris can be obtained by hydration of gypsum.
(b) Plaster of Paris is obtained by partial oxidation of gypsum.
(c) Gypsum contains a lower percentage of calcium than Plaster of Paris.
(d) Gypsum is obtained by heating Plaster of Paris.
36. Which of the following is known as fusion mixture? (1994)
(a) Mixture of Na2C03 + NaHC03
(b) Na2CO3.10H2O
(c) Mixture of K2C03 + Na2C03
(d) NaHC03
37. All the following substances react with water. The pair that gives the same gaseous product is (1994)
(a) K and K02
(b) Na and Na202
(c) Ca and CaH2
(d) Ba and Ba02.
38. Among the following oxides, the ong which is most basic is (1994)
(a) ZnO
(b) MgO
(c) Al203
(d) N205
39. Which of the following metal ions play an important role in muscle contraction? (1994)
(a) K+
(b) Na+
(c) Mg2+
(d) Ca2+
40. Which of the following statement is false? (1994)
(a) Strontium decomposes water readily than beryllium.
(b) Barium carbonate melts at a higher temperature than calcium carbonate.
(c) Barium hydroxide is more soluble in water than magnesium hydroxide.
(d) Beryllium hydroxide is more basic than barium hydroxide.
41. Which one of the following has minimum value of cation/anion ratio? (1993)
(a) NaCl
(b) KCl
(c) MgCl2
(d) CaF2
42. Which of the following has largest size? (1993)
(a) Na
(b) Na+
(c) Na–
(d) Can’t be predicted.
43. Which compound will show the highest lattice energy? (1993)
(a) RbF
(b) CsF
(c) NaF
(d) KF
44. Strongest bond is in between (1993)
(a) CsF
(b) NaCl
(c) both (a) and (b)
(d) none of the above.
45. Electronic configuration of calcium atom may be written as (1992)
(a) [Ne] 4p2
(b) [Ar] 4s2
(c) [Ne] 4s2
(d) [Ar] 4p2
46. Which one of the following substance is used in the laboratory for fast drying of neutral gases? (1992)
(a) Phosphorus pentoxide
(b) Active charcoal
(c) Anhydrous calcium chloride
(d) Na3P04
47. Compared with the alkaline earth metals, the alkali metals exhibit (1990)
(a) smaller ionic radii
(b) highest boiling points
(c) greater hardness
(d) lower ionization energies.
48. Washing soda has formula (1990)
(a) Na2C03.7H20
(b) Na2CO3.10H2O
(c) Na2C03.3H20
(d) Na2C03
49. Which one of the following properties of alkali metals increases in magnitude as the atomic number rises? (1989)
(a) Ionic radius
(b) Melting point
(c) Electronegativity
(d) First ionization energy
50. Which of the following atoms will have the smallest size? (1989)
(a) Mg
(b) Na
(c) Be
(d)Li
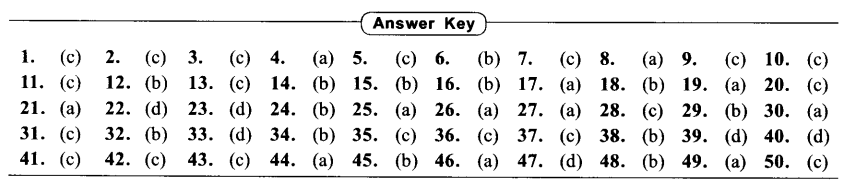
Answer Key
Explanations













Leave a Reply